Liraglutide
Premium API
Liraglutide API
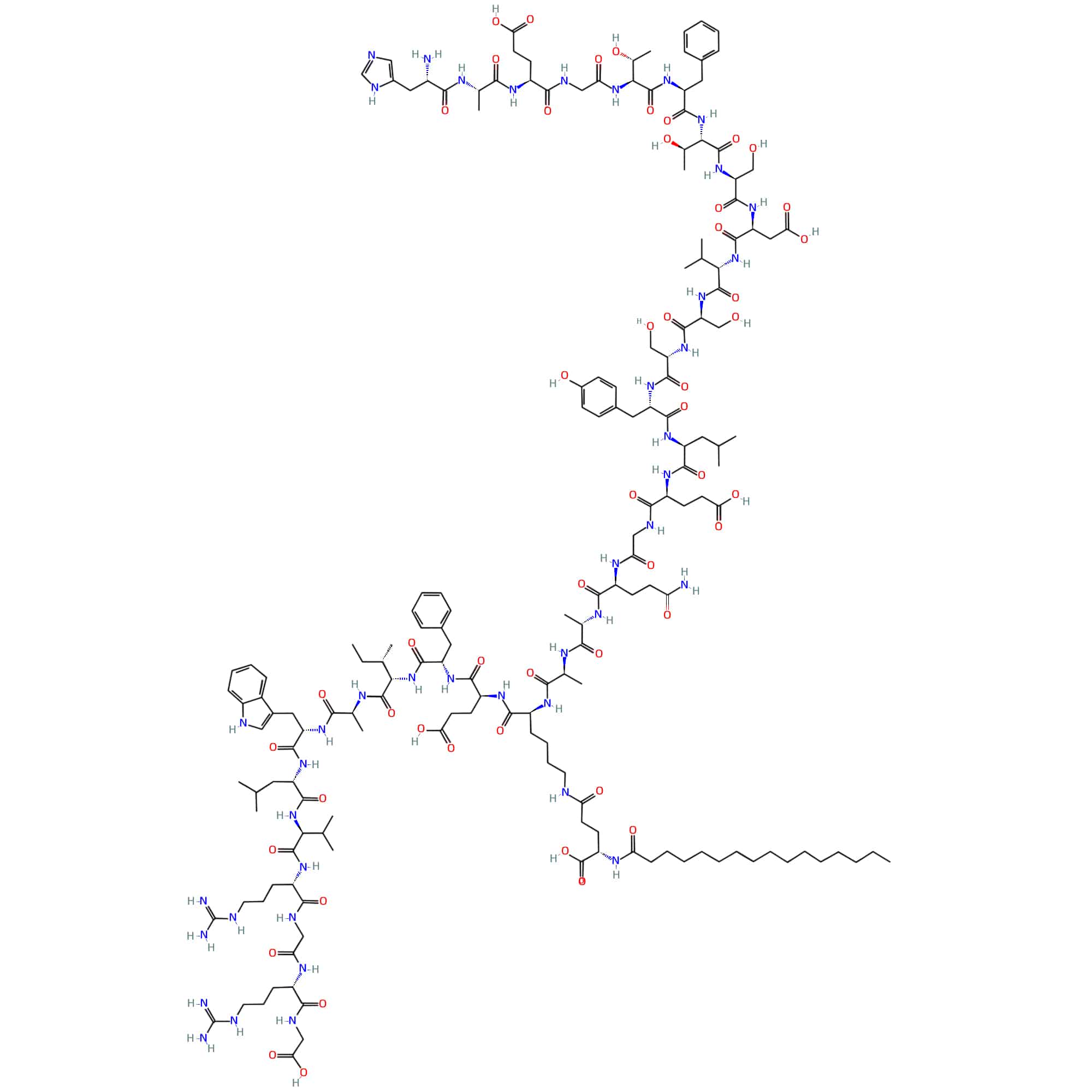
Liraglutide is a lipopeptide and polypeptide, it is a synthetic analogue of human GLP-1 with 97% similarity. The lysine residue at position 34 is replaced by arginine, and a hexadecanoyl group (palmitic acid, C-16 fatty acid) attaches to the remaining lysine at position 26 by a glutamic acid spacer. It has the functions of a glucagon-like peptide-1 (GLP-1) receptor agonist and a neuroprotective agent.

Premium API
Liraglutide
Premium API
Liraglutide Description
Liraglutide is an acylated GLP-1 receptor agonist, derived from human GLP-1(7-37), 97% similar structure of endogenous GLP-1. It contributes to insulin release in pancreatic beta cells once blood glucose increase, this insulin secretion subsides as glucose concentrations reach euglycemia (normal blood glucose level). Different from endogenous GLP-1, liraglutide is table against metabolic degradation with half-life of 13 hours.
Liraglutide is the once-daily GLP-1 derivative for type 2 diabetes treatment. The action extension of liraglutide is achieved by lipidation of GLP-1 molecule at position 26, this enable liraglutide bind reversible to albumin and release slowly over time. The effect of liraglutide is improving insulin secretion and reducing glucagon secretion in response to glucose control. Moreover, liraglutide does not affect glucagon secretion adversely, once in response to low blood sugar.
Pharmacokinetics
In reason of degradation by ubiquitous enzymes, dipeptidyl peptidase-4 (DPP4) and neutral endopeptidases (NEP), endogenous GLP-1 has plasma half-life of 1.5-2 minutes. The half-life after intramuscular injection is half an hour, this limits the usage as a therapeutic agent. The metabolically active forms of GLP-1 are endogenous GLP-1-(7-36)NH2 and rare GLP-1-(7-37). The action extension of liraglutide is achieved through attaching a fat acid molecule at GLP-1-(7-37) molecule, enable it self-associate and bind to albumin in the subcutaneous tissue and bloodstream. Then the active GLP-1 is released from albumin at slow and consistent rate.

Action Mechanism
Liraglutide is an acylated synthetic GLP-1 analog, it is an agonist of GLP-1 receptor in coupling to adenylate cyclase. Lraglutide activates the GLP-1 receptor by the stimulatory G-protein(GS) in pancreatic beta cells, it also increase intracellular cyclic AMP(cAMP) to encourage insulin release in elevated glucose concentrations. Liraglutide is also known as incretin mimetics, it increase insulin release from the pancreas and decreases excessive glucagon release to achieve glucose control function. Liraglutide is less sensitive to metabolism than the endogenous GLP-1, it is metabolized slowly by dipeptidyl pepttidase-4 (DPP-IV)and neutral endopeptidase(NEP).

our Quality
Medical Application
Liraglutide improves the control of blood sugar, it can reduce the risk for first occurrence of death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke. Liraglutide is also applied for chronic weight management.
Medicine Name of Liraglutide
Premium API
Genetic Synthesis of Liraglutide
OurLiraglutide API technology is developed by our experienced bio-engineering team.
Liraglutide is the once-daily GLP-1 derivative for type 2 diabetes treatment. The action extension of liraglutide is achieved by lipidation of GLP-1 molecule at position 26, this enable liraglutide bind reversible to albumin and release slowly over time. The effect of liraglutide is improving insulin secretion and reducing glucagon secretion in response to glucose control. Moreover, liraglutide does not affect glucagon secretion adversely, once in response to low blood sugar.
Patent Protected IP Rights:
Technology Features:

Technology Comparison and Advantages
| Morebio | Novo Nordisk | Advantages | |
| Strain-based Technology | E.coli | S.cerevisiae | – |
| Fermentation Cycle | 8 hours | 5 days | Faster production cycle |
| Process Nutrient Demand | Less | More | Cost effective |
| Target Polypeptide/Protein Yield | 5.3g/L | – | Higher than S.cerevisie-basedtechnology |
| Impurity Profile | 2 identified impurities | 3 identified impurities | Higher purity |
