Enteropeptidase
Premium Protease
Enteropeptidase
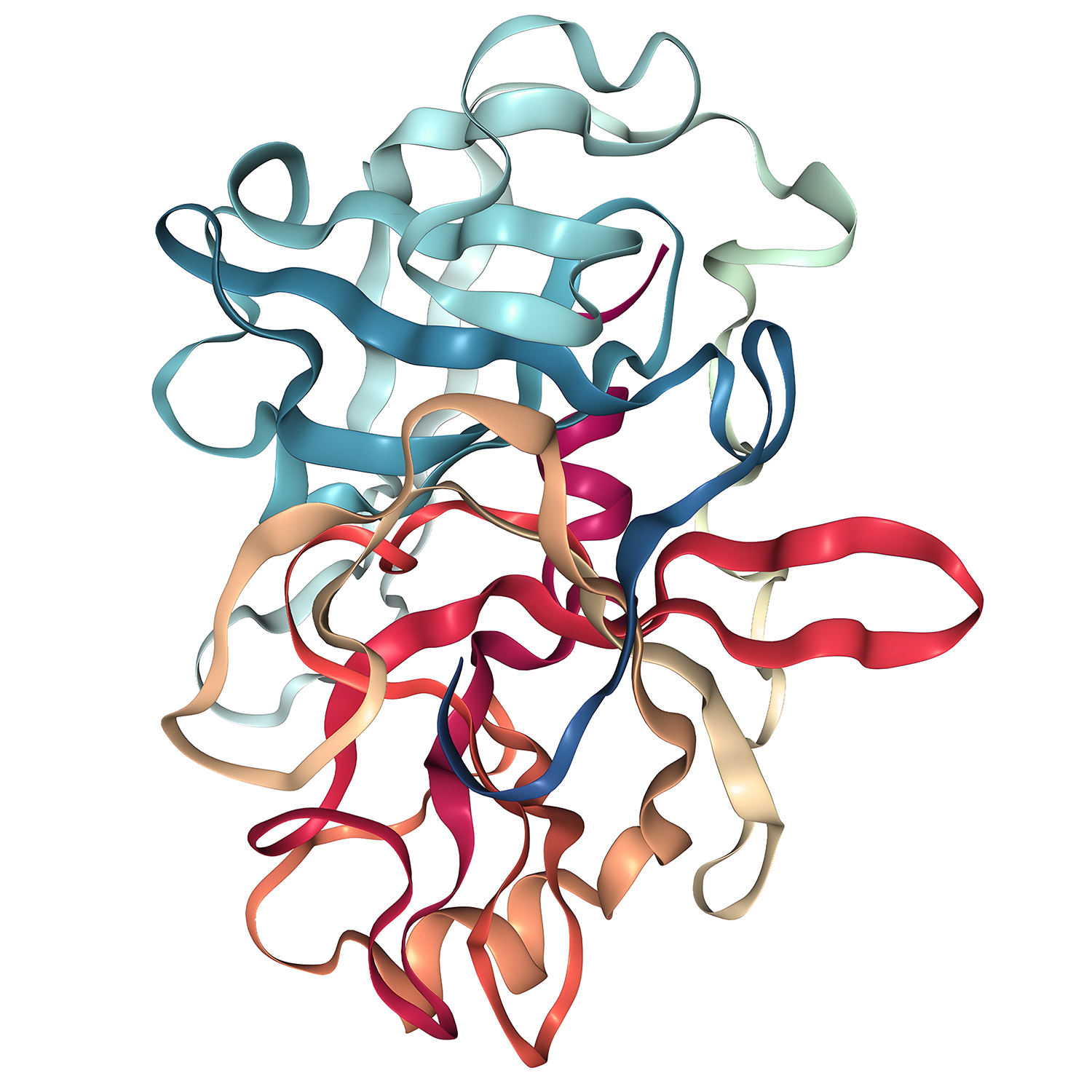
Recombinant Enteropeptidase (Enterokinase) is a high-purity, active, and specific enteropeptidase, it is produced by recombinant expression in Escherichia coli or Pichia pastoris. Recombinant Enterokinase (rEK) has the same featuring enzymatic cleavage sites as natural enterokinase, with cutting sites at Asp-Asp-Asp-Asp-Lys. This rEk can remove the fusion protein at the N-terminus, and eliminate unnecessary fusion tags.
In addition, rEK exhibits higher activity than the natural enteropeptidase, it can cleave fusion proteins effectively over a broad pH range of 4.5-9.5 and a wide temperature range. Even in various detergents and denaturing agents, it still retains partial activity.

Premium Protease
Enteropeptidase Specification
Trypsin Reference
Features
Application
Production Service
Enteropeptidase Production
Morebio develops our unique expression system of E.coli or Pichia pastoris for recombinant secretory proteins. We are the premier company to provide a professional and comprehensive microbial expression platform. We have years of experience with specific bioproducts in different microbial systems.
Custom Protein Expression
Gene Expression Workflow
Fermentation Capability
GMP Fermentation

